 |
310-206-2510
|
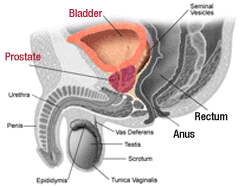
Prostate Brachytherapy
High Dose Rate (hdr) Brachytherapy For Prostate Cancer
Back to Top1. Introduction
| Click here for a PDF of the CET Prostate Brochure. The two forms of brachytherapy performed today for the treatment of prostate cancer are low dose rate in the form of permanent seeds and High Dose Rate (HDR) temporary implants or HDR Brachytherapy. Our physicians did hundreds of permanent seed implants before switching to HDR in 1991, for reasons explained elsewhere in this website. To date, our center has done over 3,000 HDR brachytherapy implants for prostate cancer. |
 |
Back to Top2.Prostate HDR Brachytherapy
HDR Brachytherapy can be used as the only treatment for prostate cancer or it can be used in combination with external beam radiation therapy (EBRT). When used as single treatment it is known as "HDR Monotherapy" and when used it is given with external beam it is known as "combined HDR and EBRT". The reasons to use HDR Monotherapy to combine HDR with EBRT are discussed in detail below. Basically, HDR Monotherapy is used for early or localized prostate cancer or in some cases where cancer has recurred after prior radiation therapy.Prostate cancer consists of a variety of illnesses from slowly progressive localized disease to a more aggressive cancer with a tendency to progress locally and spread beyond the prostate into lymph nodes, bones, or other organs. The T-stage (tumor extent determined by digital rectal examination or DRE), the level of the prostate specific antigen (PSA), and Gleason grade (the microscopic appearance) are the major prognostic indicators. The rate of rise of the PSA (amount of change over 1 year) and the number of positive biopsy specimens (fewer is better) are also important factors. A summary analysis of these prognostic factors helps the physician to recommend appropriate treatment for each individual patient.
Back to Top2.1 HDR Monotherapy
HDR Monotherapy is used for patients whose disease is believed to be confined to the prostate or immediate surrounding tissues (just beyond the covering or "capsule" of the prostate). Our HDR Monotherapy prostate protocol was developed in 1996 for patients who, for one reason or another, were not candidates for combined HDR and EBRT. We reasoned that if permanent seeds could be used without EBRT so could HDR. We subsequently observed that HDR Monotherapy for prostate cancer was a safe and effective treatment and thus began our HDR Monotherapy program. We have treated nearly 500 patients with HDR Monotherapy and it is now a routinely recommended treatment option at CET. Our results have shown excellent clinical control of disease and few side effects.
Our protocol is designed to minimize side effects by giving treatment in a series of two "implant" procedures, performed approximately one week apart. Three HDR treatments (called "fractions") are given with each implant so a total of six fractions completes the course of treatment. Since the radiation is delivered quickly, the acute radiation reactions come and go quickly. The tumor control rate (patient without clinical evidence of disease or PSA progression) is more than 95%.
Back to Top2.2 Combined HDR Brachytherapy and External Beam Radiation Therapy
Combined HDR brachytherapy and EBRT is used for patients with locally more advanced disease within or around the prostate, those with higher PSA levels or higher pathology (Gleason 8-10) grade. Delivery of EBRT expands the area of treatment beyond the prostate into other regions of the pelvis where microscopic deposits of disease may be present but undetected. Combined HDR and EBRT therapy patients undergo two HDR implants a week apart with two HDR radiation treatments (also called fractions) per implant for a total of four HDR treatments. Approximately 2 weeks later a short (4-5 weeks) course of EBRT (Monday through Friday) is given.
Our published results on patients followed for 10 years are among the best reported in the literature. We have shown 90% clinical control of disease and high rates of freedom from PSA progression (a rising PSA) that vary according to the severity of disease.
Back to Top2.3. Benefit of HDR Brachytherapy
The advantages of HDR brachytherapy for prostate cancer are:- Organ Preservation (structure and function)
- Accuracy and precision of radiation dose delivery
- Knowledge of radiation dose before treatment is given
- Ability to shape the radiation dose to fit the tumor and avoid normal tissue
- Fewer urinary, rectal and sexual side effects
- No radiation source (seeds) migration into other organs
- No radiation exposure to other people
- Short course of treatment measured in days to weeks rather than months (as with permanent seeds or external beam)
- Excellent coverage of microscopic extension of cancer immediately beyond the prostate
- Minimizes areas of radiation overdose (hot spots) or underdose (cold spots) within the prostate
- Organ motion (target movement) is not an issue for HDR as it is with EBRT
- It can be an effective treatment (termed "salvage")for cancer recurrence
- Combined HDR brachytherapy offers several other advantages over the 7-8 week course of EBRT without HDR. It offers the best opportunity to control the distribution of radiation within and around the prostate (dosimetry). Control rates are correspondingly high and complication rates low with HDR brachytherapy. Simultaneous with the best dosimetry the HDR and EBRT treatment course is shorter (more accelerated) than EBRT alone. The radiobiologic advantages (as well as convenience) to shorter treatment courses in the treatment of prostate cancer are now well known.
- HDR Monotherapy has the shortest course (1 week) and is the most conformal (matches the desired target) of all forms of radiation.
3. Effectiveness of HDR Brachytherapy
Back to Top3.1 Effectiveness of Monotherapy
The 5 and 8 year outcomes of High Dose Rate (HDR) Monotherapy for low and early intermediate risk prostate cancer are 96%.Methods
Between January 1996 and December 2003, 117 consecutive patients (median age 64 years) were treated with a series of 2 HDR implants to minimize injury to normal tissue. The procedures were performed approximately 1 week apart to allow recovery of normal tissue effects of treatment. Three doses of HDR (6.75-7.25 Gy) were delivered during each implant. The risk group characteristics were T1c (82%) and T2 (18%), Gleason 6(94%) and 7(6%), PSA <10(84%) and 10-20(16%). The risk group profile was low in 75% and intermediate in 25% of cases. The total HDR dose was escalated from 36 to 43.5 Gy over the course of our experience, since we observed excellent tolerance to therapy. The planning volume was calculated at least 5mm beyond the prostate capsule in order to encompass possible microscopic extensions of disease. The posterior margin or treatment varied according to the proximity of the anterior rectal wall to minimize side effects. The normal tissue dose constraints (limits) were anterior rectum 80% (of the prescription dose), bladder 85%, and urethra 108%. Median follow-up duration was 2 years and the mean was 2.5 years. Three different definitions of PSA progression were studied. They were the ASTRO definition (3 consecutive rises), two consecutive rises of at least 0.5 ng/ml, and nadir +2 ng/ml. Morbidity analysis was based upon RTOG criteria.
Results
The Kaplan Meier PSA progression free survival rates are identical at 5 and 8 years (0.96 ASTRO, 0.94 nadir + 2, and 0.98 two rises of >0.5 ng/ml). All but one patient had clinical disease control. Upon further follow-up, the two patients who were ASTRO definition failures did not have PSA progression. In other words, the PSA elevations were transient (PSA bounce) and not true failures. The cause specific survival was 100%. That means no one has died of prostate cancer. The Radiation Therapy Oncology Group (RTOG) grade 1-4 scale for measuring late morbidity revealed minimal GI side effects. Only one patient, who had ulcerative colitis before therapy, had grade 1 rectal symptoms. The late urinary morbidity was 2 grade 2, 4 grade 3 and zero grade 4. The grade 3 cases consisted of 1 patient who underwent urethral dilation for a stricture, 2 patients who performed self-catheterization, and 1 patient had a laser transurethral resection for benign prostate hypertrophy (BPH).
Conclusions
The CET HDR Monotherapy protocol is safe and effective therapy for low and early intermediate risk group prostate cancer.
HDR monotherapy reliably delivered the dose to the prostate target, known as the clinical treatment volume (CTV). The therapeutic dose known as the planning treatment volume (PTV) was delivered to a 5 mm margin beyond the prostate and it included part of the seminal vesicles closest to the prostate. The dose at the prostate capsule was approximately 110% of the target dose. The dose to the rectum, urethra, and bladder were given as prescribed.
Advantages of HDR Monotherapy
There appear to be fewer side effects with HDR monotherapy than with combined HDR and EBRT. A formal comparative statistical study is underway to confirm our impression. Doses to both the target and nearby tissues are known before radiation given and there is no radiation exposure to other persons. The treatment is accurately delivered in minutes with the patient and implant in the same position as when the planning images were obtained. The implant is checked with radiographs or fluoroscopy before each treatment to ensure catheter position is correct. HDR reliably delivers the dose to a prescribed distance beyond the prostate capsule and there is no concern about shifting or migrating seeds. The ability to control the HDR dwell times (the location and time the source spends at each location within the implant) is thus like having a supply of infinitely variable strength seeds to insert at precisely measured positions within the prostate. The dose to 90% of the prostate (called the D90 for permanent seeds) is always 105-120% with HDR. The volume of the prostate covered by the 100% dose (V100) is always 97% or better. A small volume (2-3%) is purposely given slightly lower doses to protect the urethra where it enters and leaves the prostate. Thus, unlike the permanent seeds literature, the measures of HDR dose always demonstrate complete coverage of the target and excellent protection of the normal organs.
Back to Top3.2 Effectiveness of Combined Therapy
Our long term results have been published in the International Journal of Radiation Biology and Physics. Vol. 61, No. 5, pp. 1306–1316, 2005 under the title: HIGH-DOSE-RATE INTENSITY-MODULATED BRACHYTHERAPY WITH EXTERNAL BEAM RADIOTHERAPY FOR PROSTATE CANCER: CALIFORNIA ENDOCURIETHERAPY’S 10-YEAR RESULTS D. Jeffrey Demanes M.D. and others.
The article describes a series of 209 consecutive patients treated with combined HDR brachytherapy and EBRT during years 1991 and 1998. The median follow-up was 7.25 years. None of the patients received androgen deprivation therapy (hormone therapy). The patients were divided for analysis into three risk groups:
Low (Stage T2a or less, Gleason score <6, and PSA <10 ng/mL)
Intermediate (Stage T2b or T2c, Gleason 7, or PSA 10–20 ng/mL)
High (Stage T3, Gleason score 8–10, and PSA level >20)
Four definitions of PSA progression were assessed and then compared for evidence of clinical failure. The rate of general clinical control for the entire group was 90% (188 of 209). The overall survival rate was 79%, and the cause-specific survival rate was 97%. The PSA progression free survival (ASTRO definition) rate was 90% for low, 87% for intermediate, and 69% for high-risk groups. The rate of late urinary morbidity was 6.7 Grade 3 was 1% Grade 4. There was no late Grade 3 or 4 rectal morbidity. The sexual potency preservation rate was 67%.
These 10 year results show that HDR and EBRT is safe and effective treatment for all stages and risk groups of prostate cancer and that the CET method compares favorably with other therapeutic alternatives.
Long Term Results With and Without Androgen Deprivation
We have presented the results of a study that compares the outcome of combined HDR and EBRT when a short course of androgen deprivation therapy was also used. It asks the question of whether androgen suppression improves the outcome with HDR brachytherapy and it also doubled the number of patients studied (with long term follow up) treated with the HDR and EBRT at California Endocurietherapy. The results have been presented at national meetings: 1) American Brachytherapy Society (ABS), San Francisco 2005 2) American Society for Therapeutic Radiation and Oncology, Denver 2005
LONG TERM RESULTS OF HIGH DOSE RATE BRACHYTHERAPY AND EXTERNAL BEAM WITH AND WITHOUT ANDROGEN SUPPRESSION FOR PROSTATE CANCER: D. Jeffrey Demanes, M.D., Gillian Altieri, C.M.D., David Brandt, M.A., Marie-Claire Barnaba, MSN., Pat Skoolisariyaporn M.S.., Lionel Schour, M.D. and Dennis R. Hill, M.D.
We reported the long term outcome of High Dose Rate (HDR) brachytherapy and external beam radiation therapy (EBRT) with and without short-term (6 months) neoadjuvant and concomitant androgen deprivation therapy (ADT).
Methods
Between Sept. 1991 and Dec. 1998, 411 consecutive patients were treated with 4 fractions HDR (5-6G) and 36-39.6Gy (the dose was increased over time as we observed that side effects were low). The study was prospective, but it was not randomized for androgen deprivation therapy (ADT). It compared patients treated with the same HDR and EBRT protocol with ADT as the only known variable. There were 200 patients in the "ADT group" (20% low, 48% intermediate, and 32% high risk) and 211 in the “no ADT "(33% low, 44% intermediate, 23% high risk). A comparison of treatment parameters revealed that patients who received ADT were more likely to be in a higher risk group. However, we divided patients into risk groups (called stratification) for analysis to balance the major prognostic factors. Also whole pelvic EBRT was given to 20% of the "ADT group" and 1.5% “no ADT group” (although other studies of HDR have shown no benefit to treatment of the whole pelvis compared to prostate only radiation therapy. The ASTRO definition of failure was used to define PSA progression. The median follow up was 7 years. General clinical failure end points were local or distant failure, PSA >25, or placement on androgen deprivation. General clinical control was the inverse.
Results
The ASTRO PSA progression free survival (PSA-PFS) for all patients at 8 years was 85% and by risk group: low 93%, intermediate 86%, and high 71%. Low and intermediate risk groups were not statistically different from one another but they both did statistically better than the high risk group. An overall group comparison between "ADT group" and “no ADT group” revealed no significant difference in general clinical control (89% vs. 89%), PSA-PSF (86% vs. 83%) or cause specific survival (97% vs. 97%). The clinical local control (no evidence of recurrence in the prostate) rate was 99%. PSA progression was no difference between "ADT group" and “no ADT group” despite the higher rate of whole pelvic radiation for the "ADT group".
Conclusion
HDR brachytherapy and EBRT is a safe and effective treatment for all risk groups of patients with prostate cancer. Unlike the randomized trials that use EBRT without brachytherapy, the outcome stratified by risk group was not improved by the use of androgen deprivation therapy (ADT). Long term follow up of the CET HDR brachytherapy and EBRT patients shows the PSA progression free survival for low and intermediate risk group patients to be excellent. High risk group patients also have good control rates compared to other treatment approaches. Lack of randomization in the study means that some degree of case selection bias cannot be excluded, but our results suggest further study of ADT with HDR will be required to demonstrate which risk groups of patients, if any, will benefit from ADT.
4. Who Qualifies for HDR Monotherapy and Combined HDR and EBRT
Back to Top4.1 Who Qualifies for Monotherapy
1. Stage T1c, T2a, or small T2b (limited findings on digital rectal exam)
2. PSA < 15 (as long as the PSA doubling rate is > 1 year)
3. Gleason 2-7 (3+4 not 4+3 and it involves < 50% biopsy specimen)
The above criteria are applied to patients on an individual case basis. Most patients with low risk group disease (T1c or T2a, Gleason 6 or less, and PSA <10) will probably be candidates for HDR monotherapy. Some patients categorization as intermediate risk group will be also considered candidates. For example, a patient with Gleason score of 3+4=7, a small palpable tumor (T2a or T2b), and a PSA < 15 would be consider a candidate for HDR monotherapy, if a only small percentage of the biopsy specimen is positive and the MRI indicates a low volume of disease and no extracapsular extension.
Certain patients with prior radiation (external beam or permanent seeds) who have biopsy proven evidence of persistent disease may be candidates for "salvage" HDR monotherapy.
Back to Top4.2 Qualifies for Combined HDR Brachytherapy and EBRT
All patients and risk groups are eligible for the combination HDR and EBRT because the long term results have demonstrated both safety and efficacy in all risk groups. Patients who have locally recurrent disease after prostatectomy may be evaluated for one of the HDR prostate brachytherapy programs depending upon the interval since the prostatectomy, the original risk group status, and other factors.Back to Top5. Side Effects
The possible post-HDR acute side effects, usually lasting two to four weeks, are urinary frequency, dysuria, and urinary retention. There may also be some usually transient rectal irritation or penile numbness (10% cases). The perineal area will be tender for a few days so activities like bicycle riding that put pressure on this area should be avoided. Medications are prescribed to help alleviate these temporary side effects.Back to Top6. HDR Brachytherapy Procedure
- The physician determines what course of treatment the patient should follow after reviewing the patient's history and recent test results. This is discussed during the consultation. The usual time between consultation and the first implant is 3 to 4 weeks. The day before the implant, the patient comes to the clinic for a pre-op exam and blood test.
- In the OR, the patient is given spinal anesthesia, which numbs the patient from the waist down. The patient's legs are placed in stirrups and the pelvis is positioned by the physician for access to the perineum (the space between the scrotum and anus).
- CET physicians use a non-fixed template technique for placing the treatment catheters, called '"flexiguides'" around and through the prostate. The rubber template has pre-drilled holes arranged in two concentric circles, rather than the square grid arrangement of the fixed template technique. Our physicians feel the circular distribution conforms better to the natural shape of the prostate. The template is held in one hand, while the flexiguides are inserted with the other. Holding the template, rather than having it clamped to the table as in the fixed template technique, allows the physicians to tip and angle the template, resulting in more control over where they can place the flexiguides. Our physicians can flare the flexiguides to encompass any size prostate, perineural or extracapsular disease or seminal vesicle involvement that may be present. This ability to flare the flexiguides enables our physicians to treat larger prostates without having to increase the number of flexiguides, which is 17. The pubic bone can sometimes interfere with the placement of rigid steel needles used in permanent seed implants, but using the non-fixed technique and plastic flexiguides, the pubic bone poses much less of a problem. This means that the patient doesn't have to take pre-implant hormones in order to shrink the gland, a common practice if permanent seeds are used.
- Once the flexiguides are in position, the template is sutured to the perineum to stabilize the implant. A radio-opaque catheter is placed in the bladder for urine drainage and urethral visualization. A rectal catheter is inserted to allow liquid contrast to be inserted into the rectum for radiographic visualization. After a brief stay in the recovery room, the patient is brought to our clinic for radiographic imaging (films and/or CT). Once the images of the implant are obtained, the patient waits for the customized treatment plan to be generated.
- After the physician has approved the treatment plan, the patient is moved into the treatment booth, where the protruding ends of the catheters are connected to transfer tubes, which are in turn connected to the afterloader. Although the patient is alone in the treatment booth, he is constantly monitored via closed circuit TV and intercom. The patient will hear the afterloader click and whir as the source is directed into each of the flexiguides. The Iridium-192 source delivers the radiation dose in about 10 minutes. After the treatment, the source withdraws into the afterloader. The patient is no longer radioactive.
 |
Fig 1: This patient is receiving his treatment. The afterloader is in the lower right corner. | |||
- The patient goes to his hospital room for the overnight stay. He is allowed to roll from side to side, but not sit upright as that would place pressure on the protruding ends of the flexiguides. The next morning, the second treatment is given This second treatment requires much less time because the simulation and treatment planning procedures do not need to be done again. However, a pre-treatment fluoroscopy is done to make certain that the implant hasn't moved during the night. After the second treatment (or third treatment if the patient is having HDR monotherapy), the template and treatment flexiguides are removed. During the removal process, there is some slight bleeding, which is stopped by the nurse or therapist by applying firm pressure to the perineal area with gauze pads. The patient goes back to his hospital room where the nurses make sure that the patient can urinate before going home.
- The second implant is done a week later, following the same procedure as described above.
7. Treatment Planning (Dosimetry)
Back to Top7.1 3D CT Based Dosimetry
One of the great advantages of HDR over permanent seeds is the ability to evaluate the implant in 3-D and know what the doses are going to be before any treatment is given. The ability to control the time the source spends in each dwell position allows for much greater target conformality and normal tissue sparing.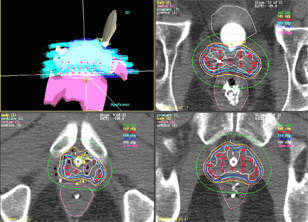 |
Fig 2: These four images are used to evaluate the implant using the 3D CT based software. The upper left image shows the implant in 3D. The physician can rotate the implant to assess the radiation coverage by the prescription dose "cloud" from any angle, and the proximity of the implant to other structures, such as the rectum and bladder. Clockwise, three CT images show the radiation dose levels at the top, middle and bottom of the gland. | ||
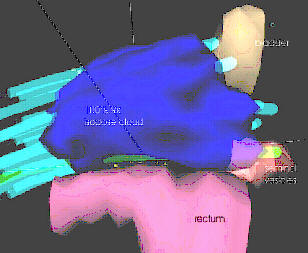 |
Fig 3: This close-up 3-D image of the 100% isodose cloud, showing near complete coverage of not only the prostate plus 5mm margin, but also the proximal seminal vesicles, shown semi-transparently. Notice how the dose cloud is shaped posteriorly to spare the anterior rectal wall. | ||
 |
Fig 4: This view evaluates the implant from the top. The red areas are the small portions of the target volume that are spared the full prescription dose: the bladder neck and the lower bulbous urethra to prevent urinary incontinence and urethral stricture respectively. | ||
Back to Top7.2 Dose Volume Histrogram
Another way of evaluating the implant is to plot a dose volume histogram (DVH). A DVH tells the physician how much of the target volume is covered by the 100% prescription dose, and how much of the prescription dose the rectum and urethra receive. At CET, we routinely see 95% or higher, coverage to the target volume, which includes the proximal seminal vesicles and a 5mm margin around the prostate to include any extracapsular extension. The actual prostate receives 10% to 25% higher than the prescription dose. This results in a D90 (the dose 90% of the prostate volume receives) of 105 to 113% compared to most permanent seed implants which are considered 'optimal' if the D90 is 90%.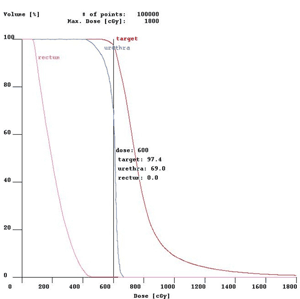 |
Fig 5: This DVH shows that 97.4 % of the target volume is being encompassed by the prescription dose. 35% of the urethra received doses slightly higher than the prescription dose, with the maximum dose being around 110% of the prescription dose. The maximum anterior rectal wall dose our physicians allow is 75% of the prescription dose, versus up to 100% with seeds or external beam. | ||
Back to Top7.3 Radiograph Based Dosimetry
There are some instances where CT cannot be used to do the dose calculations. Hip prostheses cause artifacts in the CT images making the prostate and catheters hard to identify. Patients with severe claustrophobia have a problem being scanned. This technique, while not resulting in 3-D images, is a highly accurate technique that has been used for years before CT imaging became available.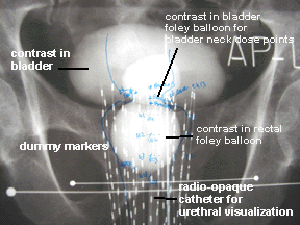 |
Fig 6: This is an "AP" (front view) radiograph of a CET prostate implant. The blue markings are the rectal outline and dose calculation points for the bladder, urethra and rectum. "Dummy" markers are inserted in the catheters. Contrast is introduced into the bladder (cystogram). Contrast is also used to inflate the foley balloons in the bladder and rectal catheters. The bladder catheter is made of a radio-opaque material to see the urethra clearly. Notice how the catheters can be made to flare in order to encompass any extracapsular extension. | ||
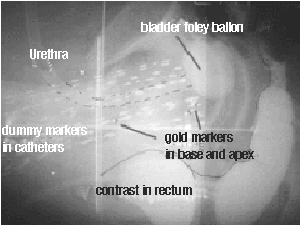 |
Fig 7: This is a lateral (side view) of the CET prostate implant. The physicians try to lift the prostate away from the rectum to help achieve low rectal doses. Notice how the tips of the catheters are against the bladder. This ensures that any tumor in the base of the gland is adequately treated, yet by adjusting the time the source spends in the tip "dwell" positions, the bladder doses are controlled. | ||
Back to Top7.4 Typical CET HDR Dose Distributions
Back to Top7.5 Tumor Specific BoostTypical CET HDR Dose Distributions
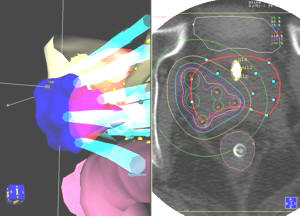 |
Fig 9: Specific Radiation Boosting: CET was the first center to realize and use the ability of the HDR system to send the source to specific positions in the catheters allowing us to give an additional "boost" dose to the cancer nodules. The image on the left shows the 3-D boosted area in blue at it extends out past and into the (red semi-transparent) prostate. The transverse view CT image with the isodose levels is on the right. | ||
Composite Dose Distribution
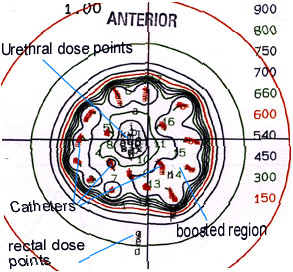 |
Fig 10: Composite Dose Distribution | ||
| This is the resulting isodose pattern of the initial whole gland plan combined with the boost, which we call the composite plan. The numbers are the isodose values in cGy (HDR). The highest dose areas are in the peripheral zone of the prostate, where most prostate tumors arise. Notice the larger boosted area that is covered by the 900 cGy (HDR) isodose line. The prescription dose, 600 cGy (HDR), is the inner red line going around the prostate. Note that the 600 cGy prescription is occurring 5mm out from the prostate capsule. If necessary, we can create an | |||
Back to Top7.6 Example of HDR Conformity and Accuracy: Sparing a TURP defect
A common procedure done to relieve pressure of an enlarged prostate on the urethra is called a transurethral resection of the prostate (TURP). An incision is made in the urethra and part of the urethra and prostate tissue is removed. Some men experience some incontinence after this procedure. In order to avoid exacerbating this situation, we spare the TURP defect as much as possible by limiting the radiation dose to less than 105% of the prescription dose. We have found that this dose is tolerated by the urethra while still delivering the necessary tumoricidal dose to the prostate.The HDR system allows for much greater conformality than permanent seeds. With seeds, one hopes that they don't move near or into the defect and the dose to the defect is not known after the one month post-implant dosimetry is done. With HDR, we can accurately shape the radiation dose to avoid the defect while knowing what the doses will be before any treatment is given.
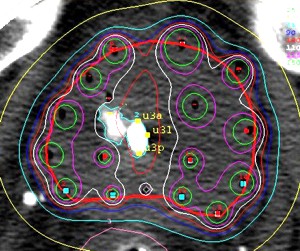 |
Fig 11: HDR Conformity and Accuracy Sparing a TURP defect: This image shows how we can accurately shape the radiation dose (white line) to go around the contrast-filled TURP defect. This precision is not possible with EBRT or permanent seeds. | ||
Home
Treatment Programs
Back to TopGeneral Frequently Asked Questions
1. What is Brachytherapy?
The prefix "brachy" is the Greek word for "short" distance. Brachytherapy is a form of internal radiation treatment where radioactive sources are placed on or into cancer tissues. There are two kinds of brachytherapy. The radiation sources may be inserted either permanently or temporarily. The two most common forms of treatment are low dose rate (LDR) permanent seeds for prostate cancer and high dose rate (HDR) temporary brachytherapy, that can be used for prostate, gynecologic, breast, head and neck, lung, esophageal, bile duct, anorectal, sarcoma, and other cancers.
2. What is high dose rate (HDR) Brachytherapy?
High dose rate (HDR) is a technically advanced form of brachytherapy. A high intensity radiation source is delivered with millimeter precision under computer guidance directly into the tumor killing it from the inside out while avoiding injury to surrounding normal healthy tissue. For a more in depth explanation please visit the understanding HDR Brachytherapy page.
3. How does radiation kill cancer?
Cancer is made of abnormal cells that tend to grow without control. Cancer DNA is more sensitive to radiation than are normal cells, so radiation kills cancer directly or when the cells attempt to multiply while normal tissue in the region is able to repair and recover.
4. What are the advantages of HDR Brachytherapy?
- Short course of treatment compared to other types of radiation treatment (1 week)
- Preservation of organ structure and function
- Fewer side effects
- Excellent coverage of possible microscopic extension of cancer
- Knowledge of radiation dose distribution before treatment is given
- Accuracy and precision of tumor specific radiation dose delivery
- Minimizes areas of radiation overdose (hot spots) or underdose (cold spots)
- Organ motion (target movement) is not a problem for HDR as it is with external beam
| Prostate Specific |
- Effective treatment for cancer recurrence (termed "salvage" therapy)
- No radiation source (seeds) migration into other organs
- No radiation exposure to other persons
| Breast Specific |
- Conserves the breast and yields excellent cosmetic results
- Reduces radiation dose to the heart, lungs, and opposite breast
- Doesn't cause a delay in other treatments such as chemotherapy
For more information on the advantages for specific cancer sites please click on the appropriate link below:
Prostate cancer | Breast Cancer | Gynecologic Cancer | Head & Neck Cancer
Esophageal and Bile Duct Cancer | Lung Cancer | Soft Tissue Sarcoma Cancer
5. How successful is HDR Brachytherapy?
HDR Brachytherapy is effective treatment of local disease in many forms of cancer including prostate, gynecological, breast, head and neck, esophagus, lung, anorectal, bile duct, sarcoma, and other primary cancer or localized metastasis as reported in medical literature. CET's publication on prostate cancer, for example has demonstrated 90% 10-year tumor control. Success rates for other tumors vary according to the type and stage of cancer being treated.
6. How many treatments has CET administered?
As of 7/31/2009, CET has performed 10,090 HDR implants and delivered 21,428 HDR treatments. Please see our treatment statistics for further details.
7. Why is HDR less well known than other forms of cancer treatment?
HDR Brachytherapy is a relatively new form of advance radiation technology. Fewer physicians have been trained to perform HDR procedures compared to seed implants or external beam radiation. Few centers, other than CET have been dedicated to the development of HDR brachytherapy to its full potential. Dr. Demanes has devoted his career to the advancement of brachytherapy and has pioneered the use of HDR and established CET as a center of excellence with specially trained and experienced staff and physicians.
8. Why should I select CET?
- Most experienced HDR center in the country
- First center specializing solely in HDR brachytherapy
- Recognized as HDR experts by colleagues in radiation oncology
- Acknowledged safety record
- Highly trained and experienced physicians and staff
- Long term results published in peer reviewed medical literature
- Quality patient care and follow up
Back to TopAbout Us
-
California Endocurietherapy Cancer Center (CET) is the first brachytherapy only center in the United States.
-
Founded by D Jeffrey Demanes M.D. in 1981.
-
Dedicated solely to High Dose Rate brachytherapy (HDR) since 1991.
-
Most experienced HDR brachytherapy center.
-
A training destination for physicians and residents.
-
HDR treatment protocol development
-
Innovation in high dose rate brachytherapy and equipment design
-
Dedicated to long-term follow-up, outcome studies, and publications in medical journals.
 |
 |
 |
| American Society for Therapeutic Radiology And Oncology Chair - Health Policy and Economics Practice Management Subcommittee, Chair - Regulatory Subcommittee, Member - Health Policy and Economic Committee, Member - Health Policy and Economics Code Development and Valuation Subcommittee, Member - Code Utilization and Application Subcommittee. American Brachytherapy Society Chair - Socioeconomic Committee. American College of Radiation Oncology President - 2005 to 2007 American College of Radiology Fellow - 2007 |